Mitigating Risk and Improving Quality of Care
Co-authored by Lorianne Maria Sainsbury-Wong, Esq., CISSP, CIPP/US, CHPC and Joanne Byron, BS, LPN, CCA, CHA, CHCO, CHBS, CHCM, CIFHA, CMDP, COCAS, CORCM, OHCC, ICDCT-CM/PCS
This paper outlines the basics related to key steps, metrics, and best practices for implementing an effective CDI audit program. The information below is for educational purposes only and not intended as consulting or legal advice.
Introduction
Clinical Documentation Improvement (CDI) is a vital process that ensures medical records are accurate, complete, and compliant, directly impacting patient care quality, severity-of-illness tracking, and reimbursement. As CDI departments mature, establishing a robust, routine auditing process—both internal and external—is essential to validate the accuracy of CDI staff queries, identify educational gaps for physicians, and ensure compliance with regulatory standards.
Effective CDI audits identify gaps in diagnostic specificity, medical necessity, and coding accuracy which must support documentation (like histories and exam findings), and appropriate, non-leading queries.
OIG Guidance and Regulatory Support for Your Audit
To control risk, your internal auditors will benefit from a clearer understanding of healthcare compliance guidance, statutory and regulatory provisions that are related to CMS reimbursement. The HHS Office of Inspector General’s (OIG) General Compliance Program Guidance (2023) calls for a proactive approach that emphasizes preventing errors rather than relying on post-submission rationalizations. The OIG guidance implicates several key documentation safeguards: documentation must accurately reflect the services provided; patient records must be unique to the specific clinical encounter documented; and an effective risk mitigation playbook should address systemic documentation errors before they lead to overpayment demands or other matters.
Grounded in statutory frameworks, Title XVIII of the Social Security Act sets forth a principle of “no documentation, no payment.” All diagnostic and therapeutic interventions must meet the “reasonable and necessary” standard. Medical records that provide insufficient information to justify the conditions for CMS payment could result in claim denials or a subsequent recoupment of funds.
Checklist for Clinical Documentation Improvement Audits
A CDI audit is a structured review designed to measure the effectiveness of the CDI program in capturing the full clinical picture of a patient. It serves as a check-and-balance system, evaluating not only the accuracy of coding but also the appropriateness of queries sent to providers. A CDI audit also ensures that the medical record reflects real-time clinical practices rather than functioning as a retrospective cost justification.
The process involves a continuous, four-stage cycle which should have a Lead Auditor to guide the team to: 1) Prepare and plan (set goals), 2) Execute – collect and analyze records, validate findings, 3) Report audit findings 4) Provide education to implement change, and re-audit to ensure changes are sustained.
1. Preparing for the Audit
Success in auditing requires careful planning and preparation.
- Define Scope and Goals: Identify specific areas of focus, such as high-risk diagnoses (e.g., sepsis), high-volume, or high-cost areas.
- Select Samples: Utilize a representative sample of records, including those with queries and those without, to assess both CDI activity and documentation gaps.
- Determine Audit Frequency: Establish a regular schedule (e.g., monthly or quarterly).
- Identify Reviewers: Use a mix of internal staff for ongoing monitoring and external auditors for unbiased, independent assessments.
2. Execute the Audit
An effective CDI audit follows a standard quality improvement cycle (Plan, Do, Study, Act):
- Data Collection
Use random sampling of patient records to get a representative view or targeted sampling for specific providers or types of documentation. Reviewers gather patient records, specifically examining:- Specificity to ensure documentation is accurate, thorough, and detailed.
- Medical necessity - Confirm that the documentation justifies the care provided.
- The principal diagnosis assigned.
- Secondary diagnoses (comorbidities and complications).
- Present on Admission (POA) indicators.
- Query compliance, ensuring queries are evidence-based and not leading, with best practices focused on clarifying ambiguities in the medical record
- Analysis and Validation
Auditors compare the documentation against evidence-based standards. Key questions include:- Did the documentation support the queried diagnosis through objective clinical indicators, e.g., vitals, labs, treatment?
- Was the query necessary, or was the information already in the record?
- Are there missed opportunities where documentation was insufficient?
3. Reporting Audit Findings
Audit findings should be reported through actionable metrics.
- Query response rates and agreement percentages. Report if providers respond to queries and if those queries improve the record.
- DRG shifts (change in Diagnosis Related Group).
- Denial reduction rates.
4. Provide Education to Implement Change
The results are used to provide targeted feedback to clinicians and CDI staff.
- Develop educational sessions based on recurring documentation gaps (e.g., chronic condition management).
- Update templates and checklists for improved accuracy.
- Re-audit to ensure improvements are sustained.
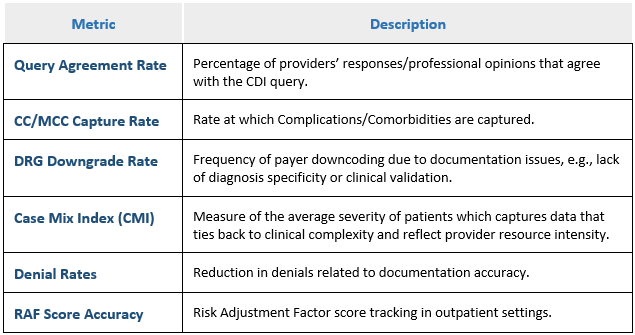
Key Metrics/Key Performance Indicators (KPIs) to Audit
To measure the success of a CDI program, organizations should track specific key performance indicators (KPIs):
Conclusion
Best practices for successful CDI audits involve the providers. After all – it is their documentation being audited! Ensure the CDI team creates processes that minimize administrative burden.
Leverage technology and streamline the audit process by using computer-assisted coding (CAC) and AI-powered analytics to scan for gaps and prioritize reviews. Ensure documentation is accurate across all patient encounters, not just for higher reimbursement.
Share feedback. Collaborate with the coding and billing departments to ensure documentation aligns with ICD-10/CPT guidelines. Create a closed feedback loop where findings are shared with clinicians and coders for ongoing training.
Remember, auditing for CDI is a continuous cycle of improvement, moving beyond simply chasing revenue to establishing a sustainable, compliant, and accurate record-keeping process. By focusing on regular reviews, actionable metrics, and ongoing education, organizations can improve the quality of clinical documentation, leading to better patient care and optimal financial outcomes.
About the Authors
Lorianne Maria Sainsbury-Wong, Esq., CISSP, CIPP/US, CHPC, is a member of the AIHC Volunteer Education Committee. Joanne Byron, BS, LPN, CCA, CHA, CHCO, CHBS, CHCM, CIFHA, CMDP, COCAS, CORCM, OHCC, ICDCT-CM/PCS, is the Chief Executive Officer at the American Institute of Healthcare Compliance.
References
American Institute of Healthcare Compliance
National Library of Medicine
Office of Inspector General, U.S. Department of Health and Human Services. (2023). General Compliance Program Guidance.
Social Security Act § 1815(a), 42 U.S.C. § 1395g(a)
Social Security Act § 1862(a)(1)(A), 42 U.S.C. § 1395y(a)(1)(A)
